This FAQ was co-developed by primary and specialty care to support the assessment and management of osteoporosis in adult patients within the primary care setting. It is intended to be used in conjunction with specialty advice services, such as Endocrinology tele-advice for complex or non-urgent cases. For tele-advice services, click here.
-
What do I include in the secondary workup for osteoporosis after Dual-Energy X-ray Absorptiometry scan (DXA) and Fracture Risk Assessment Tool (FRAX)?
Consider in everyone:
Creatinine (GFR)
Calcium
Phosphate
ALP (alkaline phosphatase)
TSH
SPEP (if vertebral fractures)
25-OH vitamin D
Consider based on symptoms:
Workup for Cushing’s: urine free cortisol or 1 mg dexamethasone suppression test
Workup for multiple myeloma: SPEP, Urine protein electrophoresis (UPEP), free light chains
Workup for hypercalciuria: 24-hr urine calcium and creatinine (especially if suspicion of malabsorption)
Workup for celiac: anti-TTG
Workup for hypogonadism in men: total testosterone (+/- LH, FSH, prolactin)
Workup for hypogonadism in women: menstrual history
Workup for inflammatory disorders: Erythrocyte Sedimentation Rate, RF, ferritin, total iron-binding capacity test, tryptase, histamine, homocysteine, skin biopsy (only as history dictates)
Occult fractures: T and L spine lateral x-ray
There are a few basic labs to consider in all patients in whom diagnosis and treatment of osteoporosis is considered. Additional tests should be guided by the history and physical exam.
The recommended biochemical testing for secondary causes of osteoporosis and for potential limitations when considering pharmacotherapy are available in the Supplementary Materials section of the 2023 Osteoporosis Canada Guidelines.
The purpose of these labs is to ensure there isn’t another cause driving the low bone density/high fracture risk (i.e., secondary osteoporosis) and to ensure there are no contraindications to pharmacologic therapy.
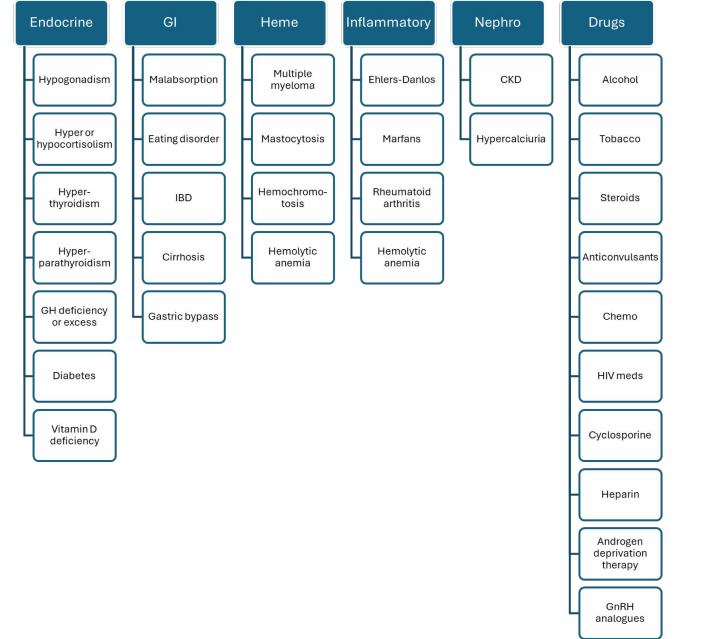
Common secondary causes of osteoporosis include hyperparathyroidism (high calcium, low phosphate), hyperthyroidism, hypogonadism, chronic inflammation, multiple myeloma, malabsorption and chronic kidney disease.
Osteomalacia is impaired mineralization of the bones and can mimic osteoporosis but has very different treatment. This is part of the reason to check calcium, phosphate, and vitamin D.
Common contraindications to pharmacologic therapy for osteoporosis include renal dysfunction, profound vitamin D deficiency and hypocalcaemia.
Note that the guidelines suggest “calcium corrected for albumin.” Routine correction for albumin in a well outpatient is no longer recommended.
Note that routine use of bone turnover markers is not recommended.
Note that routine PTH testing is not recommended. This test should be reserved for further workup of hypercalcemia.
-
What workup is required in a young person?
Most osteoporosis fractures in pre-menopausal women and younger men are due to secondary osteoporosis. Therefore, the focus in young people is on identifying and treating underlying risks.
Peak bone mass is achieved by the second or third decade; conditions that onset earlier in life can compromise peak bone mass, resulting in low BMD.
The role of DXA (Dual-Energy X-ray Absorptiometry scan) in young people is controversial. BMD does not correlate well with fracture risk in younger people and there is disagreement about definitions of osteoporosis.
BMD ordering is restricted to patients >50, and this makes good sense as BMD does not correlate as well with fracture risk in younger people. The Osteoporosis Clinic is NOT able to order a BMD on your behalf; if you believe there is a true underlying bone disorder, send a referral. If the question is for a patient-requested “baseline BMD” or there is uncertainty about a fracture, consider ruling out secondary causes.
Consider genetic causes in young people without secondary causes.
Consider suboptimal peak bone mass: full history focusing on medical conditions up to mid-late 20s when peak bone mass is achieved (systemic illness, medications, eating disorders, etc.).
The diagnosis of osteoporosis in young people is tricky—FRAX requires age >40 and BMD does not correlate well with fracture risk so a thorough history, physical exam and lab investigations are required.
Typical investigations: Ca, Mg, Phos, PTH, Vitamin D, alkaline phosphatase, LH, FSH, estradiol/testosterone, electrolytes, creatinine, liver, SPEP, celiac, TSH, A1c
Consider: tryptase, 24-hour urine calcium, 24-hour urine cortisol, Insulin-like growth factor 1 , hepatitis serology, HIV
Secondary causes:
![]()
-
How do I order a bone mineral density study for a patient less than 50 years old?
The regulations in Alberta only permit ordering of a bone mineral density (BMD) study in a patient less than 50 years old if organized by specific subspecialists including those physicians with: Endocrinology/metabolism, gastroenterology, general surgery, internal medicine, nephrology, orthopedics, pediatric (including sub specialties), physical medicine and rehabilitation or rheumatology.
In pre-menopausal women, the normal reference range for BMD encompasses Z-scores between +2 and −2, as Z-scores reflect age-matched comparisons to the population average. T-scores, which compare BMD to peak bone mass, are not clinically meaningful in this population and should not be used for interpretation. Importantly, a low Z-score in a pre-menopausal woman does not predict fracture risk and does not indicate osteoporosis in the same way it does in post-menopausal women. -
What if there is a discordance between hip and lumbar spine t-scores?
Discordance between the lumbar spine and femoral neck t-scores can be encountered in clinical practice. It should be noted that lumbar spine t-scores should not be included in the bone mineral density (BMD) section of the Fracture Risk Assessment Tool (FRAX) as it is calibrated for a femoral neck t-score.
In general, you can increase or decrease FRAX estimate for a major osteoporotic fracture by 10% (1/10th) for each rounded t-score difference between the lumbar spine and femoral neck.
For example, a patient has a t-score at the femoral neck of -2.6 and their major osteoporotic fracture risk using the FRAX risk assessment is 17%. If their lumbar spine t-score is -3.6, their major osteoporotic fracture risk would be adjusted up (given the lumbar spine t-score is worse than the femoral neck t-score) to 18.7%. Conversely, a patient has a t-score at the femoral neck of -3.6 and their major osteoporotic fracture risk using the FRAX risk assessment is 19%. If their lumbar spine t-score is -2.4, their major osteoporotic fracture risk would be adjusted down (the femoral neck t-score is worse than the lumbar spine score) to 17.1%. -
When should I initiate a drug holiday?
What is a drug holiday?
A drug holiday is the intentional, temporary interruption of drug therapy for a specific clinical purpose.
This only applies to bisphosphonates. We must not do a drug holiday on denosumab as there is a risk of rebound vertebral fracture.
Bisphosphonates accumulate in the bone and release over time, so there is ongoing bone protection long after the medication has been stopped.
What is the purpose of a drug holiday?
No added benefit: studies suggest that after a prolonged exposure to bisphosphonate therapy, there is little added benefit from ongoing treatment. Bisphosphonates are not lifelong medications, unlike many other chronic disease treatments.
Potential for harm: the rare but serious side effects of osteonecrosis of the jaw (ONJ) and atypical femoral fracture (AFF) increase with time on therapy. Those risks decrease when the medication is stopped.
In response to the wear and tear of everyday life, our bones are constantly remodelling. Remodelling requires both osteoclastic activity (bone breakdown) and osteoblastic activity (bone building). Bisphosphonates inhibit osteoclasts, so excessive bisphosphonate exposure can impair normal bone maintenance processes.
Therefore, the purpose of a drug holiday is to balance the benefits of bisphosphonate therapy (fracture risk reduction) with potential harms (ONJ and AFF).
When do we consider drug holiday?
Reassess risk after three-five years of oral bisphosphonate therapy (most studies have looked at 5 years of oral bisphosphonate use, so this is more common in clinical practice) or three years of IV zoledronic acid.
How do I decide about a drug holiday?
Like all osteoporosis treatment decisions, the choice to initiate a drug holiday is a balance of risks and benefits. Therapy can be continued in people who remain at high risk of fracture and discontinued in people who are not at high risk.
The literature is quite vague about what constitutes ongoing risk. Most people were at high risk if they were started on therapy. Consider the following factors in determining risk:
Patient factors that increase risk:
Age
Female sex
Low BMI
Ongoing co-morbidities or medications (e.g., glucocorticoids)
Falls
Hip or vertebral fracture on therapy
Medication factors:
Less potent bisphosphonate (e.g., risedronate)
Less time on therapy (either shorter duration or missed doses or improper administration)
Investigations:
Ongoing decline in BMD
Practically, if a patient has completed five years of oral bisphosphonate (with good technique and timely administration) or three years of IV bisphosphonate, has had no new fractures, BMD is stable, and no major changes in health or medications, then we can offer a drug holiday.
If there has been a decline in BMD, new fractures, or new information that elevates risk (e.g., new diagnosis, new medication) then therapy can either be continued (additional 5 years of oral or 3 years of IV) or changed (e.g., switch from oral to IV bisphosphonate).
What do we do on holiday?
Continue non-pharmacologic management: dietary calcium, supplemental vitamin D, weight-bearing exercise
Avoid things that are harmful to the bones: smoking, excess alcohol, steroids, falls
Bottom Line
Optimal duration of bisphosphonate therapy is not known
Bisphosphonates should not be continued lifelong (unless limited life expectancy)
Re-evaluate risk/benefit of bisphosphonate therapy after three-five years
Initiate drug holiday if good exposure to medication, no new fractures, no added clinical risk factors, stable BMD.
-
When do I restart medications after a drug holiday?
Why would I restart medications?
Osteoporosis is a chronic condition. We lose bone with age (about 1% per year), so fracture risk increases with age.
Bisphosphonates slowly dissociate from bone over time, losing their protective effect over years.
Does it matter what the holiday was from?
While all bisphosphonates lower fracture risk, they have different binding affinities and potencies.
Overall, zoledronic acid lasts the longest, followed by alendronate, then risedronate.
When should I reassess risk?
In most cases, you can repeat a BMD after 3 years of drug holiday.
You can re-evaluate clinically at any time, generally recommended one-two years after stopping risedronate, two-three years after stopping alendronate or zoledronic acid.
If a patient is at higher risk, consider evaluating sooner. If very low risk, consider a longer holiday.
How do I reassess risk?
Same process as before! Fracture history, FRAX, BMD, etc.
When do I restart therapy?
New fracture, change in clinical risk factors (e.g., FRAX is now >15-20%), decline in BMD >1-2%/year.
The decision needs to be individualized. We expect most patients to have multiple courses of bisphosphonate therapy throughout their lifetime.
What do I restart?
There is very little data about fracture outcomes in subsequent courses of therapy. Some experts suggest a full course again, others a shortened course.
You can consider restarting the same therapy or switching medications. There is no known “lifetime exposure” maximum for bisphosphonates, and if an adequate drug holiday of several years has been undertaken, we would start again at 0 in counting time on treatment.
Bottom Line
Many patients will need another course of bisphosphonate therapy after a several year drug holiday.
The duration of drug holiday depends on patient factors (age, co-morbidities, fall risk, etc.) and medication factors (shorter drug holiday for risedronate compared to alendronate and zoledronic acid).
While there is no clear data to guide decisions, consider restarting therapy if there has been a new fracture, FRAX >15-20%, or BMD decline >1-2%/year or T <-2.5.
-
Can denosumab be stopped?
Denosumab should not be stopped abruptly. Delays of more than one month or sudden discontinuation can result in rapid bone loss and rebound vertebral fractures in the following year in up to 8-10% of cases. The degree of bone loss is thought to be related to duration of therapy and may be highest in those with a history of prior vertebral compression fractures. Strict adherence to dosing of denosumab is essential.
If discontinuation of denosumab (Prolia®) or its biosimilar (Jubbonti®) is planned, current guidelines recommend the preferred strategy would be transitioning to zoledronic acid 5 mg IV, administered six months after the last denosumab dose, with a second zoledronic acid infusion one year after the final denosumab dose. Consider referring to a specialized osteoporosis clinic if there are questions around transition off denosumab. In these cases, specialized bone turnover markers such as c-telopeptide, measuring osteoclast (bone breakdown) activity may be used to guide transitioning off denosumab.
The optimal duration of denosumab therapy is unknown. Published data support use for up to 10 consecutive years. Longer-term use may be associated with rare adverse events (e.g., atypical femur fracture, osteonecrosis of the jaw), similar to long-term bisphosphonate therapy.Before initiating denosumab, a clear plan should be established regarding treatment duration and transition strategy. The anticipated one–two years of zoledronic acid consolidation therapy should be factored into the total planned duration of antiresorptive treatment and patients should be counselled accordingly prior to starting therapy.
-
Do all patients on steroids need therapy?
Low
Moderate
High
Very high
FRAX MOF (%)
<10
10-19
20-30
>30
FRAX Hip (%)
<1
1-3
3-4.5
>4.5
T- score
AND >-1
OR -1 to -2.4
OR -2.5 to -2.5
OR <-3.5
OR prior fracture
OR pred 30mg+ for 30+d
Therapy
Non-pharmacologic
Consider oral bisphosphonate
Oral bisphosphonate
Consider anabolic
Scope of the problem:
Glucocorticoids can lead to significant bone loss and increase fracture risk. They have impacts on bone turnover, bone microarchitecture, muscle strength (mechanical bone loading), falls risk and calcium metabolism (less gut absorption, less renal reabsorption).
Even small doses of steroids (prednisone 2.5 mg/d) over long periods of time (>3 months) can increase the risk of fracture.
Fractures occur at higher BMD than the general population (i.e., BMD alone may not accurately predict fracture risk).
Steroids are used to treat inflammatory conditions, which themselves are known to increase fracture risk (i.e., both the therapy and the underlying disorder can be harmful to bone health).
Investigations:
Consider usual secondary workup
Baseline BMD if over age 50 and intending steroid therapy for >3 months
Non-pharmacologic management:
Avoid smoking, alcohol
Minimize falls risk
Exercise (including balance, strength, resistance, and impact training)
Minimize the dose and duration of steroids where possible
Ensure eugonadal status (replace estrogen or testosterone as appropriate)
Ensure adequate calcium and vitamin D. Some sources suggest targeting a vitamin D level >75 nmol/L although there is some uncertainty about “optimal” levels. Calcium and vitamin D alone are insufficient for fracture prevention in those at high risk.
Who to treat with pharmacologic therapy:
The decision to treat with pharmacologic options depends on age, prior fractures, BMD, FRAX, and dose/duration of steroids.
If the steroid dose is >7.5 mg prednisone, FRAX will underestimate risk, so it is suggested to multiply the major osteoporotic fracture (MOF) risk by 1.15 and that of hip fracture by 1.2.
Very high doses of steroid (>30 mg prednisone/d for 30+ days) is automatically considered high risk.
The American College of Rheumatology suggests this risk stratification for those over age 40:
Type of therapy:
Oral bisphosphonates are first line, IV zoledronic acid if unable to tolerate oral
Be cautious with denosumab as this requires an exit strategy
Anabolic therapy (most evidence for teriparatide) if very high risk
Duration of therapy:
Treat for the duration of steroids. At the end of glucocorticoid therapy, re-evaluate risk and discontinue bisphosphonate if the patient falls in the low-risk category
Younger patients:
There is less guidance for patients under the age of 40. Consider reproductive potential (try to avoid bisphosphonates in reproductive-age women). This is a more nuanced decision and is worth a call to Specialist Link or referral.
-
Do we need to hold bisphosphonates for dental work?
Why are we worried about this?
Osteonecrosis of the jaw (ONJ) is a rare complication of antiresorptive therapy (oral and IV bisphosphonates, denosumab)
ONJ is exposed bone in the jaw that does not heal after 8 weeks
ONJ is NOT TMJ—many patients will tell you about jaw joint pain, which is likely unrelated
The risk of ONJ is low (<1/100,000) but the risk is increased with invasive dental work
What types of dental work does this involve?
The greatest risk is with procedures that disrupt the mucosal barrier thus exposing bone—i.e., extractions and implants
Routine dental cleanings are likely not a concern
How do I determine the risk of ONJ?
Procedural risk
This is up to the dental care provider to determine—there are clear dental guidelines on the risks of various procedures and are beyond the scope of the MD
Patient risk
Age >65
Smoking
Diabetes
Steroid use
Anti-resorptive risks
Dose (e.g., in cancer therapies, the patient may receive a greater annual dose of bisphosphonate)
Duration (risk of ONJ increases with time on therapy)
Formulation (higher risk with IV than PO bisphosphonate)
There is no data to support imaging or bone turnover markers to quantify ONJ risk
Why not just hold the medication?
There is very little evidence that a temporary hold of an anti-resorptive lowers ONJ risk
Patients on anti-resorptive medications likely have high fracture risk, therefore interrupting therapy may not be desirable
Bisphosphonates have a very long half-life. Holding therapy for a short period of time may accomplish nothing.
Denosumab MUST NOT have a drug holiday—there is a risk of rebound vertebral fracture
Who decides?
The decision is shared between the dental provider, patient, and osteoporosis provider. We are not able to “clear” a patient for a dental procedure—rather, we can help outline risks and benefits so the patient can make an informed choice.
Before starting anti-resorptive therapy, ask about any upcoming invasive dental work—ideally complete this work, allow full healing and then start therapy.
Bottom Line
There is no clear evidence that holding bisphosphonate therapy before invasive dental work lowers the risk of ONJ. Denosumab must not be held.
Anti-resorptives do increase the risk of ONJ, especially with increased time on therapy. Some sources suggest holding the bisphosphonate for two to three months prior to invasive dental work if the duration of bisphosphonate therapy exceeds three to four years. Restart when full healing is achieved. However, quality of data for this recommendation is low.
In most cases, we do not hold bisphosphonate therapy prior to dental work.
-
How soon after a fracture can we start therapy?
Initiation of fracture-reducing therapy can occur immediately after a fracture. Evidence shows that early administration of bisphosphonates does not delay clinical or radiographic fracture healing.
Bisphosphonates do not need to be discontinued to facilitate healthy fracture healing. Early treatment also reduces the risk that patients will not be reassessed and considered for appropriate therapy initiation. If imminent anabolic therapy (teriparatide or romosozumab) is being considered as first-line treatment, initiating a bisphosphonate in the interim may attenuate subsequent bone density gains. The 2023 Osteoporosis Canada Guidelines suggest considering an anabolic agent in patients with a recent severe vertebral fracture (≥40% vertebral height loss within the past two years) or multiple vertebral fractures and a T-score ≤ −2.5.
Currently in Alberta, access to anabolic therapies (romosozumab and teriparatide) remains financially challenging and may be a barrier to initiation. If imminent anabolic therapy is being considered as first line therapy, avoid starting an antiresorptive agent in the interim as it may attenuate subsequent bone density gains on the anabolic therapy. Financial considerations and insurance coverage might play a role in the decision about which therapy to start.
Anabolic therapies can be prescribed by family physicians, and access information is available at osteoporosiscalgary.com. Referrals to a specialized osteoporosis centre may also be made for therapy initiation; however, this does not change the associated medication costs. -
What if someone fractures on therapy?
While antifracture therapies have been demonstrated to lower fracture risk, no treatment can eliminate the possibility of future fractures. A fracture on therapy therefore does not always imply a treatment failure but is an important timepoint to reassess a patient.
Expert guidance would suggest:Assess for concurrent medical conditions that can predispose patients to bone loss and increased fracture risk. For example, hyperparathyroidism (starting with a calcium), myeloma (serum protein electrophoresis, free light chain ratio), hyperthyroidism (TSH), hypophosphatemia (phosphate), and if clinically appropriate, hypercortisolism, vitamin D deficiency (25(OH)D) or malignancy. Assess if patient has had new steroid exposure or initiation of other medications known to increase fracture risk.
Assess duration of therapy: It is reasonable to consider six-12 months as a minimum duration of treatment for the anticipated fracture risk reduction. Therefore, if a patient fractures before a year on therapy, it would generally not be considered treatment failure.
Assess adherence to therapy: With oral bisphosphonates, adequate adherence, generally 70 to 80% or greater, is required to achieve the expected reduction in fracture risk. Assessing administration technique is also important. These medications should be taken on an empty stomach, without other medications and patients should remain upright for at least 30 to 60 minutes after ingestion. For denosumab, assess whether there have been delays in dosing interval.
Consider a repeat BMD to see if bone density has remained stable (expected with antiresorptive use) or dropped on therapy. This tends to be more interpretable if patients have been on therapy for three years or more as earlier may be prone to measurement artifact or random variation.
Consider a switch of therapy
If on oral bisphosphonate therapy, switching to intravenous zoledronic acid or subcutaneous denosumab could be a consideration, recognizing the data when it comes to degree of fracture risk reduction is limited.
If on oral or IV bisphosphonate or denosumab, consideration could be given to switching to an anabolic agent including romosozumab or teriparatide. It would be important to note the concerns around a direct switch to denosumab therapy given the risk of rapid bone loss with denosumab discontinuation. Consultation with a specialized osteoporosis centre may be helpful.
It is also reasonable to remain on the same therapy if all the other factors are addressed and with the understanding that these interventions reduce but do not eliminate fractures.